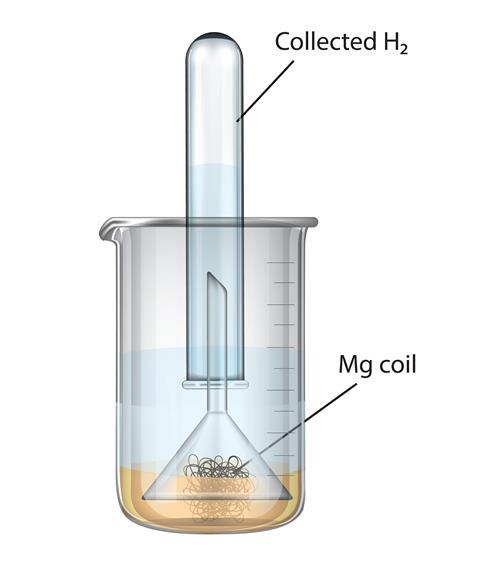
why calcium and magnesium floats on water - Science - Metals and Non-metals - 13061069 | Meritnation.com

Question Video: Balancing a Chemical Equation for the Reaction between Magnesium Nitride and Water | Nagwa
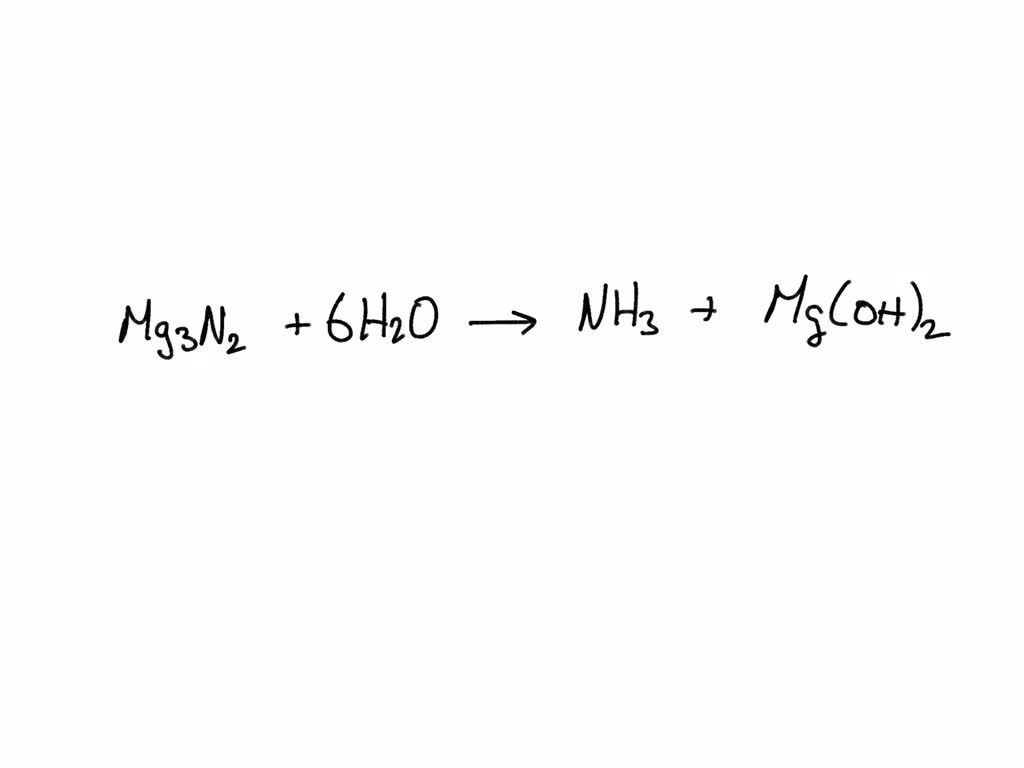
SOLVED: Magnesium nitride reacts with water to form ammonia and magnesium hydroxide. When the equation is correctly balanced, the coefficient for water is

SOLVED: (d) Magnesium sulfide reacts with water producing gaseous hydrogen sulfide and magnesium hydroxide. MgS + 2H2O -> Mg(OH)2 + H2S (i) State what would be seen during the reaction apart from

Question Video: Ordering the Reactions of Magnesium Metal with Varying Concentrations of Nitric Acid | Nagwa

Reaction of Magnesium and Water... | By Химийн лавлах | Facebook | We go across a roll on the periodic table from sodium to magnesium, the ionization energy increases and the reactivity

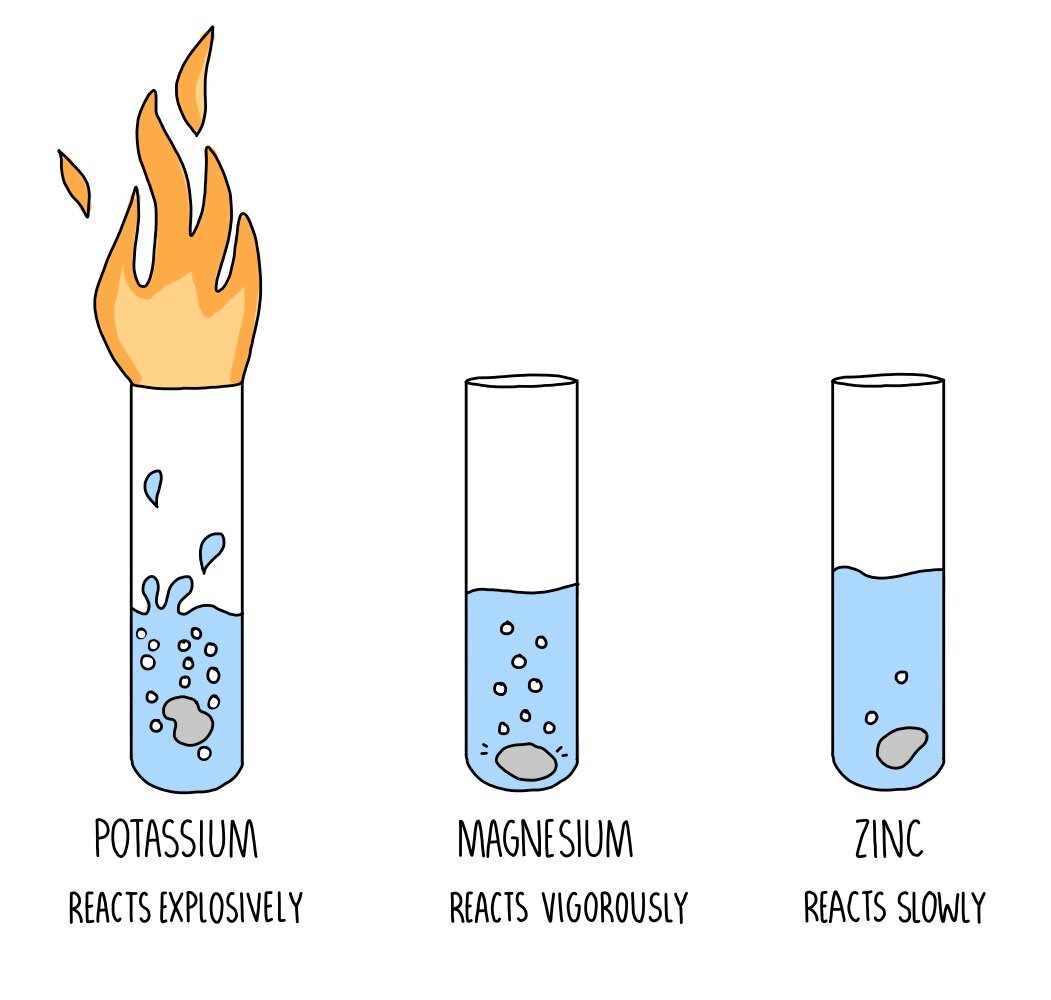
Metals react differently with water. The more vigorous is the reaction, the more is the bubble formation. The following diagram illustrates the reaction of sodium, magnesium and copper with water.What can be

science chemistry redox reaction magnesium hydrochloric acid | Fundamental Photographs - The Art of Science