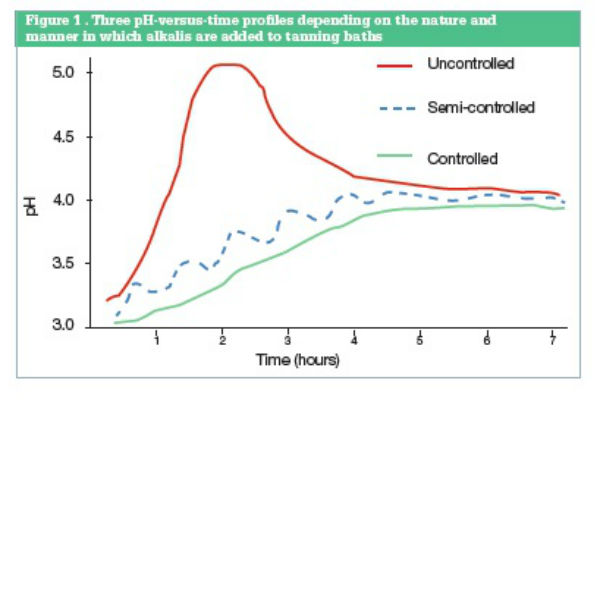
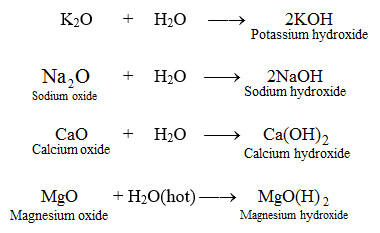Figure 1 from Magnesium bioavailability from magnesium citrate and magnesium oxide. | Semantic Scholar

A mixture consists of four substances.The properties of each of these substances are shown in the table.Magnesium nitrate decomposes on heating to form solid magnesium oxide oxygen gas and nitrogen dioxide gas.

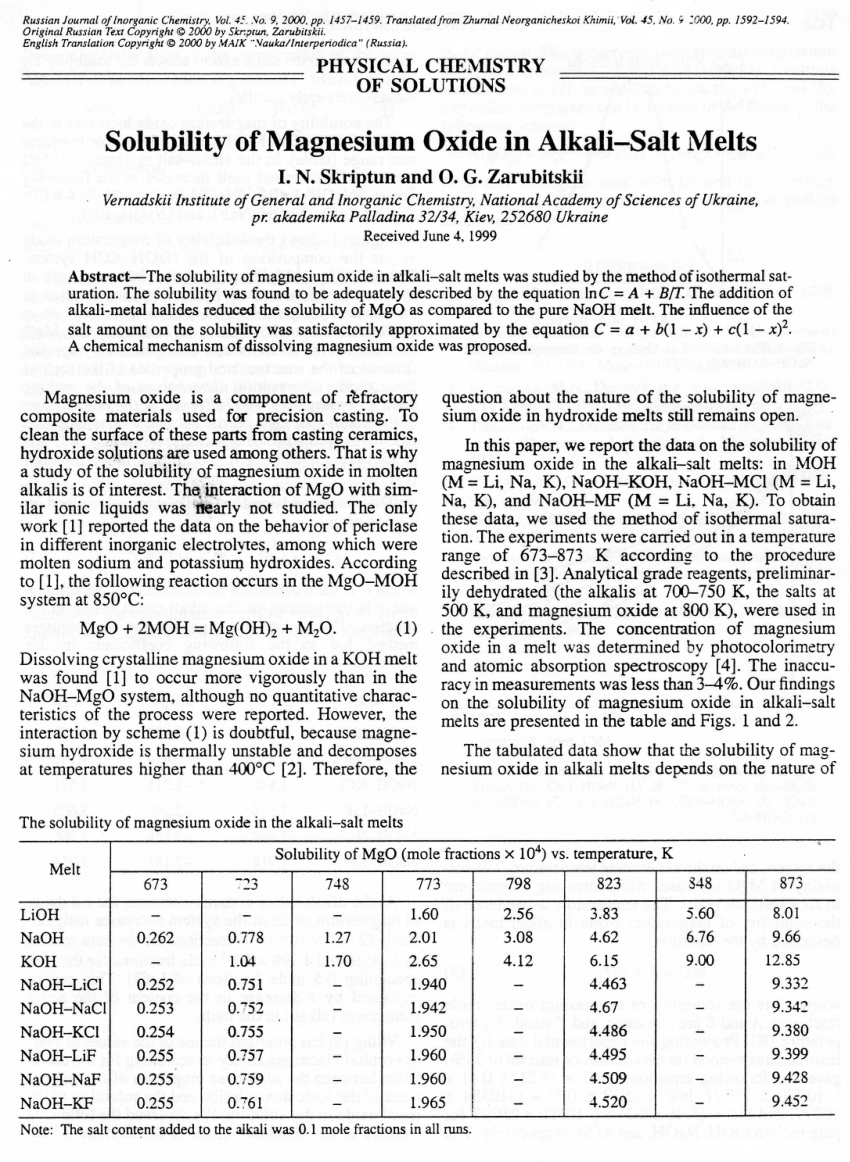
Prediction of adequate pH and Mg2+ dosage using an empirical MgO solubility model for struvite crystallization - ScienceDirect
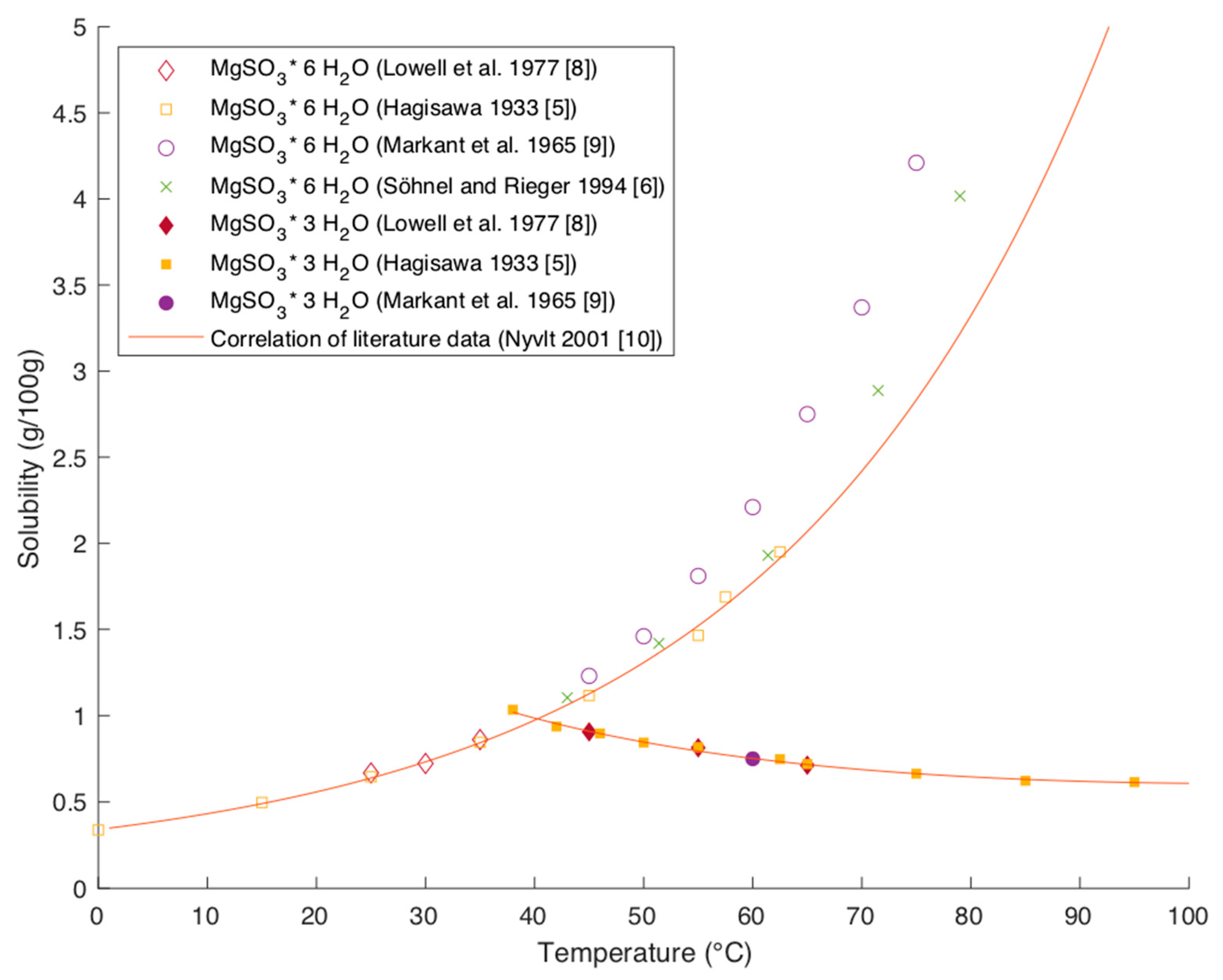
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling
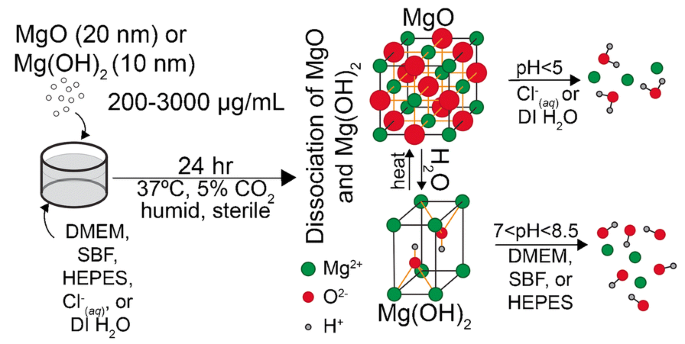
Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

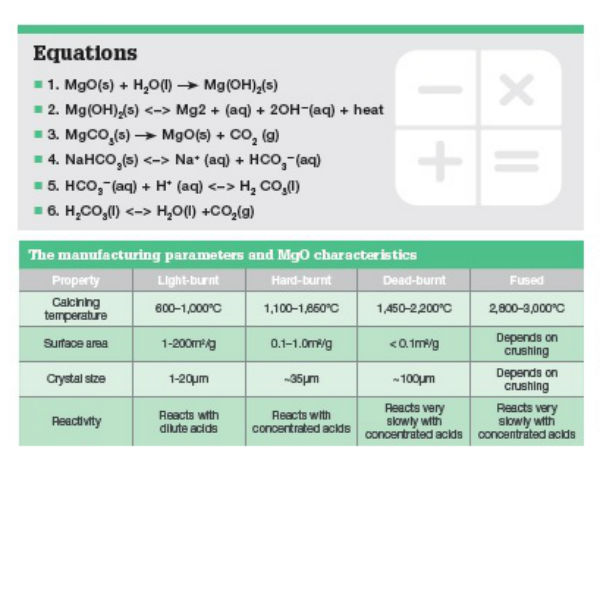
THE HYDRATION OF MAGNESIUM OXIDE WITH DIFFERENT REACTIVITIES BY WATER AND MAGNESIUM ACETATE | Semantic Scholar

Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

The solubility of magnesium sulphite hydrates calculated by Aspen Plus®... | Download Scientific Diagram