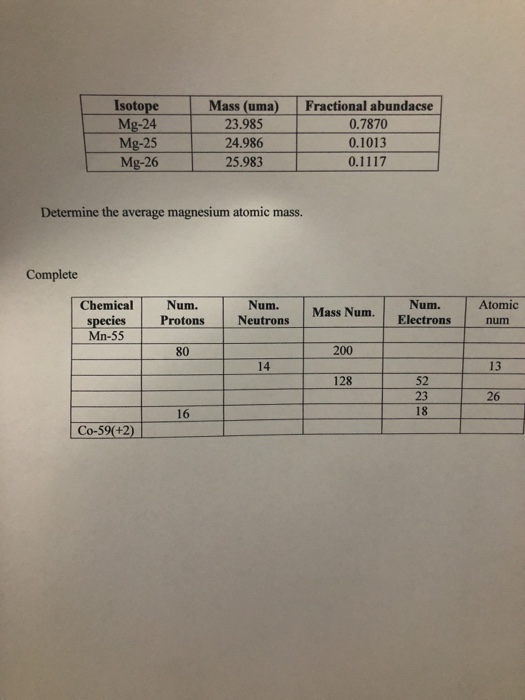
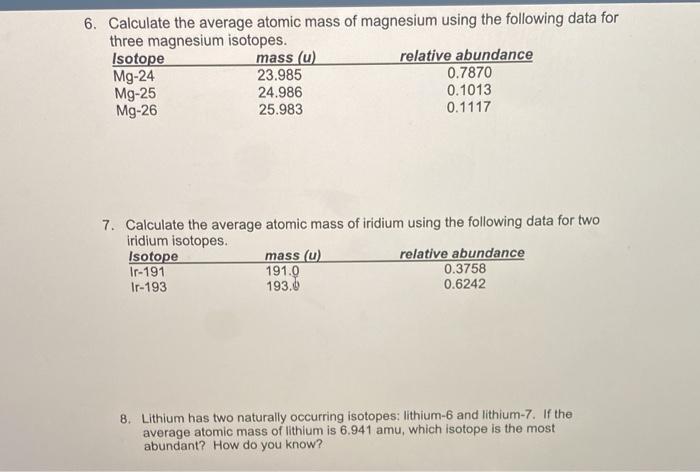
SOLVED: Calculate the average atomic mass of magnesium using the following data for threc magnesium Isotopes Ksotope massl relativg abuudance Mg-24 23.985 0.7870 Mg-25 24.986 0.1013 Mg-26 25,983 0.1417 Calculatc the average

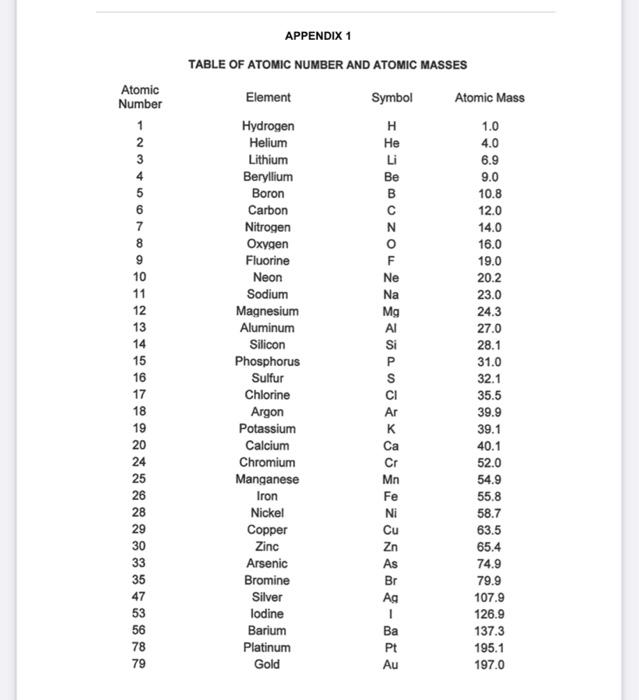
Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.

Do Now 1.What is the atomic number of copper (Cu) 2.What element has 80 protons 3.An isotope of magnesium (Mg) has a mass number of 26. How many neutrons. - ppt download
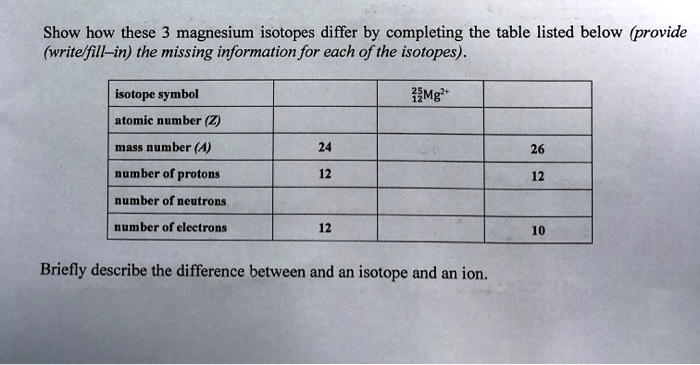
SOLVED: Show how these 3 magnesium isotopes differ by completing the table listed below (provide (writelfill-in) the missing information for each of the isotopes) . isotope symbol izMg + atomic number (2)

SOLVED: Complete the following table Name of element or ion #of neutrons #of #of protons electrons 14 12 12 140 8 Atomic Mass Symbol Magnesium Radiun 06 Mg ee Ra 226 3

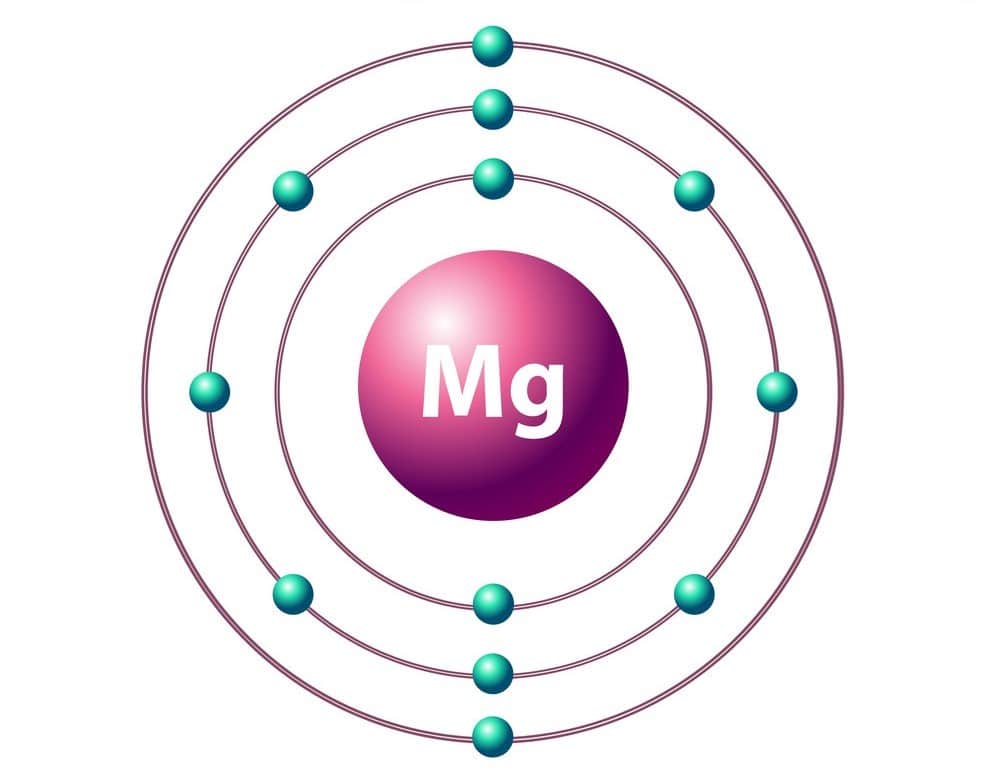
24^12Mg and 26^12Mg are symbols of two isotopes of magnesiumCompare the atoms of these isotopes with respect to: composition of their nuclei




:max_bytes(150000):strip_icc()/GettyImages-1135707671-640473b29d534e15a24491c0d6b2789e.jpg)