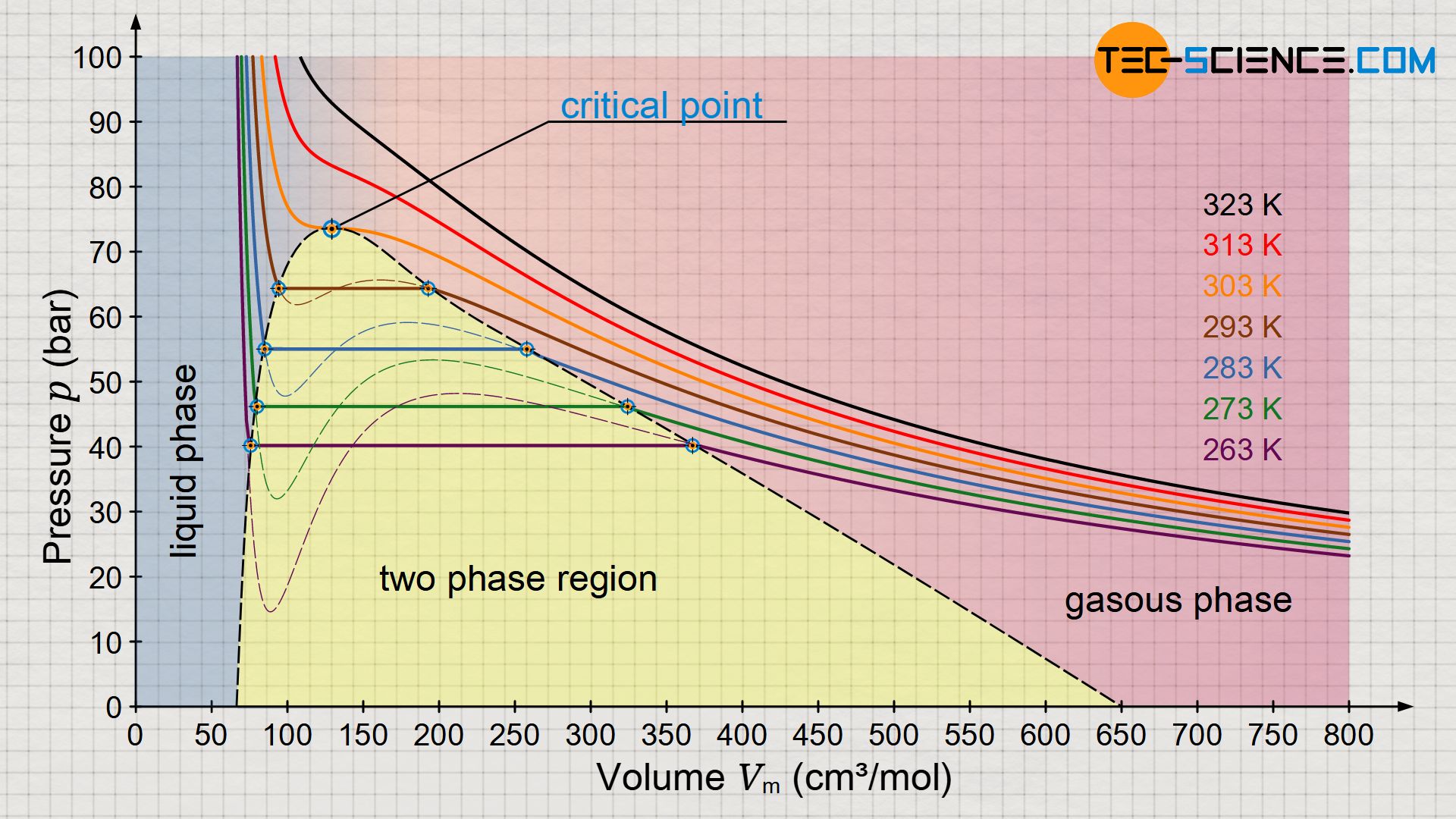
= RT , at high pressure, the Van der Waals equation gets reduced to : Using Van der Waals equation, [ P + a/V^2 ](V - b) = RT , at high pressure, the Van der Waals equation gets reduced to :](https://dwes9vv9u0550.cloudfront.net/images/9876945/d6a3d03b-a449-4d51-986d-b05943344e70.jpg)
Using Van der Waals equation, [ P + a/V^2 ](V - b) = RT , at high pressure, the Van der Waals equation gets reduced to :

SOLVED: The van der Waals equation is: P + Wo) = b RT. Where; P = pressure (atm) n = number of moles (mol) R = ideal gas constant V = volume (